
Cécile Voisset (LaTIM, groupe PRiME), Sylvain Petek, Solène Connan (LEMAR)
National
IUEM
IBSAM
IRD
Start Date
18/04/2026
End Date
Article Marine Drugs 2024
Article The Conversation 2024
LEMAR: Article du mois 2024
UBO: Actualités 2024
IRD : Brève dans IRD Le Mag’ 2025
Article Le Télégramme 2026
This project is in the spirit of the development of “Sea-Health” initiatives at UBO and inter-institute collaboration between IUEM and IBSAM. It involves 2 teams from IUEM (LEMAR and LBCM) and one team from IBSAM (UMR1078, PRiME group).
1. Scientific context :
The PrP prion protein is an endogenous protein that adopts, for unknown reasons, a pathological conformation (PrPSc) capable of being transmitted to the native form and accumulating as autocatalytic amyloid fibers. The biological mechanisms that control the propagation of these amyloid structures are still unclear. The accumulation of PrPSc causes neurodegenerative diseases such as Creutzfeldt-Jakob disease in humans, (1 person/million). There is no therapy for prion diseases that are inexorably fatal. Prion diseases are protein-folding diseases and are part of a larger group of pathologies including more common diseases such as Alzheimer’s and Parkinson’s disease. Like prion diseases, these neurodegenerative diseases are proteinopathies (protein folding diseases) in which the proteins Aβ and tau (Alzheimer’s disease) and α-synuclein (Parkinson’s disease) are found in pathological conformations.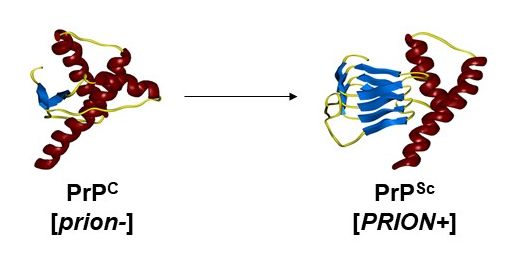
2. Objectives :
The objectives of this project are to identify marine organisms within the extract libraries (sponges, ascidians, soft corals from the Pacific ocean; macroalgae from temperate and tropical environments) of LEMAR and microorganisms from LBCM with the best potential for activity. The longer-term objective is to identify candidate anti-prion molecules that could be used for the treatment of other proteinopathies such as Parkinson’s disease.
Contributors
Cécile VOISSET, Jennifer MARTIN (LaTIM, groupe PRiME)
Yannick FLEURY, Camille JEGOU (LBCM)
Ali AL-MOURABIT, Céline MORIOU-CHESNE (ICSN-CNRS)
Mayalen ZUBIA (UMR241 EIO)
- Etudiants LEMAR: Cyril CLOAREC, Audrey BESANÇON, Colin GRUNBERGER (Alt L3, M1+2)
- Etudiants PRiME: Victorien CHAKER, Pierre COUTROT, Lucile GENTILE, Solenn BUAN, Maha SINANE (Doc)







